Chiral_Imines
(R)-2-Methyl-N-(2,2,2-trifluoroethylidene)propane-2-sulfinamide
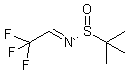
044616
In the recent decade, fluorine and fluorine-containing groups have attained a status of privileged structural tools in the design of new pharmaceuticals, agrochemicals and functional materials1. In particular, on the current pharmaceutical market about 40% of commercially available drugs contain fluorine2. With the establishment of fluorine scan as an extremely successful paradigm in the search for compounds with enhanced bioactivity and stability3, it is estimated that about 80% of new drug candidates are fluorine-containing substances. Taking into account that amino group is one of the major pharmacophores, synthesis and medicinal chemistry of amines, amino alcohols, and amino acids containing fluorine has been intensively studied4. Of particular interest are amines and amino acids derivatives containing trifluoromethyl group in alpha position to the amino function5. However, despite the tremendous amount of research data published so far4,5, there is still a need for more general, versatile, experimentally convenient methodology allowing for practical preparation of such compounds in enantiomerically pure form.
Considering the structure of the presented here compound 1, one may agree that such structural features as trifluoromethyl group, activated imine functionality and remarkably stereocontrolling6 tert-Bu-sulfinamide chiral auxiliary, render imine 1 as potential “blockbuster” chiral synthon for preparation of virtually any imaginable target alpha-trifluoromethyl containing amine or amino acid. The most obvious directions of straightforward elaboration of compound 1 may include various nucleophilic additions7 and cycloaddition reactions8. However, quite surprisingly, the chemistry of N-substituted (3,3,3)-trifluoroacetaldimines in general, and chiral derivative 1, in particular, remains significantly underdeveloped; that is in the striking contrast with the wealth of research data reported for the non-fluorinated analog6. One of the reasons for the dazzling synthetic potential of trifluoromethyl-imine 1 being still unexplored is probably its prohibitively difficult preparation as well as isolation in chemically and enantiomerically pure form. Thus most of the research reported so far7b,c,8 has been conducted using only in situ prepared imine 1; which is obviously raising the issues of experimental attractiveness and practicality.
References:
1. (a) Ma, J.-A.; Cahard, D. Chem. Rev. 2004, 104, 6119–6146; (b) Mikami, K.; Itoh, Y.; Yamanaka, M. Chem. Rev. 2004, 104, 1–16.
2. Mikami, K.; Murase, T.; Zhai, L.; Kawauchi, S.; Itoh, Y.; Ito, S. Tetrahedrom Lett. 2010, 51, 1371-1373.
3. Muller, K.; Faeh, C.; Diederich, F. Science 2007, 317, 1881–1886; and references cited therein.
4. (a) Sorochinsky, A. E.; Soloshonok, V. A. J. Fluorine Chem. 2010, 131, 127-139. (b) Mloston, G.; Obijalska, E.; Heimgartner, H. J. Fluorine Chem. 2010, 131, 829-843. (c) Kukhar, V. P.; Sorochinsky, A. E.; Soloshonok, V. A. Future Medicinal Chem. 2009, 1, 793-819.
5. (a) Liu, J.; Hu, J. Future Medicinal Chem. 2009, 1, 875-888. (b) O’Hagan, D. J. Fluorine Chem.2010, 131, 1071-1081.
6. MaryAnn T. Robak, M. A.; Herbage, M. A.; Ellman, J. A. Chem. Rev. 2010, 110, 3600–3740; 409. References in this excellent review article!
7. (a) Mimura, H.; Kawada, K.; Yamashita, T.; Sakamoto, T.; Kikugawa, Y. J. Fluorine Chem. 2010, 131, 477-486. (b) Truong, V. L.; Pfeiffer, J. Y. Tetrahedron Lett. 2009, 50, 1633-1635. (c) Truong, V. L.; Menard, M. S.; Dion, I. Org. Lett. 2007, 9, 683-685; Erratum: Org. Lett. 2007, 9, 1423.
8. (a) Dobler, M. R.; Lenoir, F.; Parker, D. T.; Peng, Y.; Piizzi, G.; Wattanasin, S. PCT Int. Appl. (2010), WO 2010031750 A1 20100325. (b) Nishimura, H.; Kawai, H.; Nitta, A.; Oyama, T.; Kumagaya, H. Jpn. Kokai Tokkyo Koho (2010), JP 2010070514 A 20100402. For general references on cycloaddition reactions of imino compounds, see: Heintzelman, G. R.; Meigh, I. R.; Mahajan, Y. R.; Weinreb, S. M. Organic Reactions, Hoboken, NJ, United States, 2005, 65.